The Concept of Equilibrium
Reversible reactions and equilibrium
- We have already seen that a reversible reaction is one that occurs in both directions
- When during the course of reaction, the rate of the forward reaction equals the rate of the reverse reaction, then the overall reaction is said to be in a state of equilibrium
Characteristics of a reaction at equilibrium
- It is dynamic eg the molecules on the left and right of the equation are changing into each other by chemical reactions constantly and at the same rate
- The concentration of reactants and products remains constant (given there is no other change to the system such as temperature and pressure)
- It only occurs in a closed system so that none of the participating chemical species are able to leave the reaction vessel
Equilibrium can only be reached in a closed vessel which prevents reactants or products from escaping system
The reaction between H2 and N2 in the Haber process
- When only nitrogen and hydrogen are present at the beginning of the reaction, the rate of the forward reaction is at its highest, since the concentrations of hydrogen and nitrogen are at their highest
- As the reaction proceeds, the concentrations of hydrogen and nitrogen gradually decrease, so the rate of the forward reaction will decrease
- However, the concentration of ammonia is gradually increasing and so the rate of the backward reaction will increase (ammonia will decompose to reform hydrogen and nitrogen)
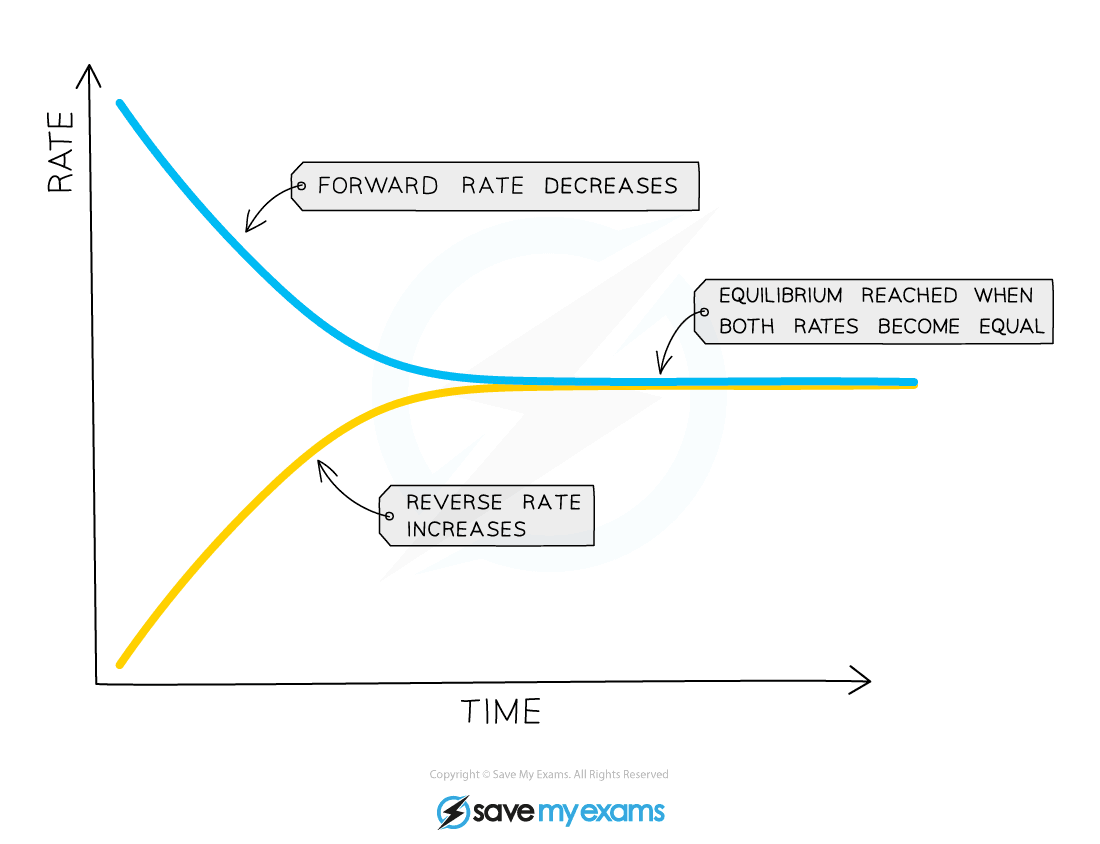
- Since the two reactions are interlinked and none of the gas can escape, the rate of the forward reaction and the rate of the backward reaction will eventually become equal and equilibrium is reached:
3H2 (g) + N2 (g) ⇌ 2NH3 (g)
Diagram showing when the rates of forward and backward reactions become equal
The position of equilibrium
- Equilibrium position refers to the relationship between the concentration of reactants and products at the equilibrium state
- When the position of equilibrium shifts to the left, it means the concentration of reactant increases
- When the position of equilibrium shifts to right, this means the concentration of product increases
Effect of catalyst on equilibrium position
- The presence of a catalyst does not affect the position of equilibrium but it does increase the rate at which equilibrium is reached
- This is because the catalyst increases the rate of both the forward and backward reactions by the same amount (by providing an alternative pathway requiring lower activation energy)
- As a result, the concentration of reactants and products is nevertheless the same at equilibrium as it would be without the catalyst
Diagram showing the effect of catalyst on equilibrium positionExtended Only
Le Chatelier’s Principle
- Le Chatelier’s Principle states that when a change is made to the conditions of a system at equilibrium, the system automatically moves to oppose the change.
- The principle is used to predict changes to the position of equilibrium when there are changes in temperature, pressure or concentration.
Effects of temperature
Example:
Iodine Monochloride reacts reversibly with Chlorine to form Iodine Trichloride
ICl + Cl2 ⇌ ICl3
Dark Brown Yellow
When the equilibrium mixture is heated, it becomes dark brown in colour. Explain whether the backward reaction is exothermic or endothermic:
- Equilibrium has shifted to the left as the colour dark brown means that more of ICI is produced
- Increasing temperature moves the equilibrium in the endothermic direction
- So the backward reaction is endothermic
Effects of pressure
Nitrogen Dioxide can form Dinitrogen Tetroxide, a colourless gas
2NO2 ⇌ N2O4
Brown Gas Colourless Gas
Predict the effect of an increase in pressure on the position of equilibrium:
- Number of molecules of gas on the left = 2
- Number of molecules of gas on the right = 1
- An increase in pressure will cause equilibrium to shift in the direction that produces the smaller number of molecules of gas
- So equilibrium shifts to the right
Effects of concentration
Example:
Iodine Monochloride reacts reversibly with Chlorine to form Iodine Trichloride
ICl + Cl2 ⇌ ICl3
Dark Brown Yellow
Predict the effect of an increase in concentration on the position of equilibrium:
- An increase in the concentration of ICl or Cl2 causes the equilibrium to shift to the right so more of the yellow product is formed
- A decrease in the concentration of ICl or Cl2 causes the equilibrium to shift to the left so more of the dark brown reactant is formed
Exam Tip
When the conditions at equilibrium are changed, the system always responds by doing the opposite.
For example if the concentration is increased the system tries to reduce it by changing the direction of the reaction or if the temperature is increased the system will try to reduce the temperature by absorbing the extra heat.