Changing State
- A change of state refers to a substance changing its state between solid, liquid or gas
The various changes of state between solids, liquids and gases
- Usually, when a substance is heated its temperature increases
- However, whilst a substance changes its state (from solid to liquid or from liquid to gas) the temperature of that material remains constant, even though energy is still being added
When a substance changes state, its temperature remains constant, even though it is still gaining (or losing) internal energy
- This happens because the energy is being used to break the bonds between the molecules instead of increasing the kinetic energy of the molecules (and hence the temperature)
- When a substance turns from gas to liquid (condensation) or from liquid to solid (solidification) the opposite happens:
The temperature remains constant whilst the bonds reform (giving out energy in the process) - The melting point and boiling point of a substance is the temperature at which that substance changes state
Boiling vs Evaporation
- Boiling and Evaporation both refer to a change in state from liquid to gas, however:
- Boiling occurs at a fixed temperature and happens throughout the liquid
- Evaporation can occur at any temperature and happens from the surface of the liquid
Evaporation occurs when higher energy molecules near the surface of a liquid escape and become a gas. This can occur at any temperatureExtended Only
Latent Heat
- Latent Heat is the energy required to change the state of a substance
- This energy is required to break the bonds holding molecules together
- (It is also given out again once the bonds reform)
- Latent Heat of Fusion is the energy required to turn a solid into a liquid
- Latent Heat of Vaporisation is the energy required to turn a liquid into a gas
- The Specific Latent Heat, L, of a substance is the energy required to change the state of 1 kg of that substance
- For an object of mass m, the total energy required to change its state is given by the equation:
Energy = m × L
Measuring Specific Latent Heat
- In your IGCSE examination you may be asked to describe an experiment to determine the specific latent heats for steam or ice
A method for determining the specific latent heat for steam is given below - In this experiment you need to use the following equation:

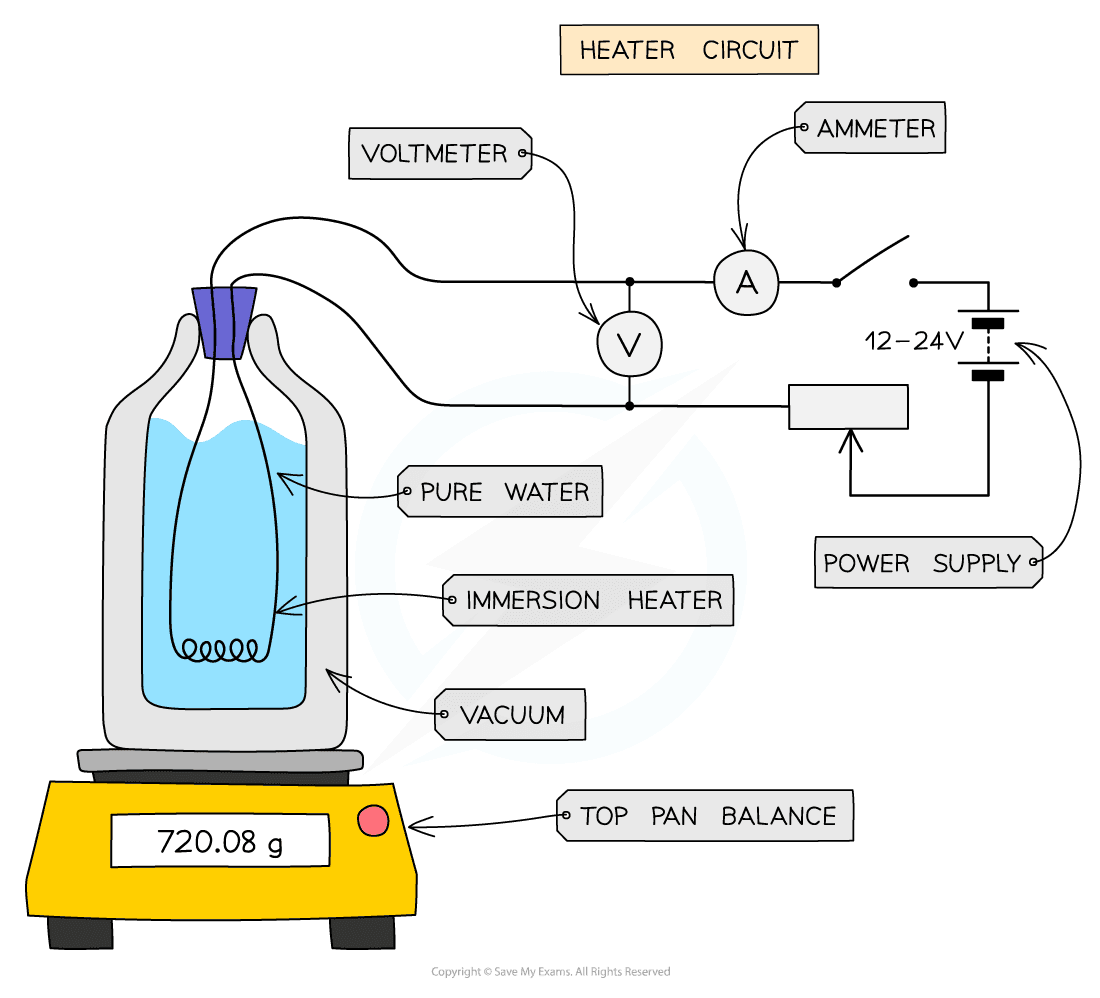
- The following apparatus will be needed:
- A vacuum flask containing pure water
- A top pan balance
- An appropriate heater (e.g. an immersion heater)
- A power source
- A joule meter or a voltmeter, ammeter and stop-clock (I will assume we have the latter)
Image showing apparatus to measure the specific latent heat for steam
- Start by measuring (and recording) the mass of the vacuum flask along with the water
- Next place the heater into the water and connect it to the power supply
- Turn on the power supply and wait until the water starts to boil
- Once the water is boiling start the stop-clock and take several measurements of the potential difference (on the voltmeter) and current (on the ammeter), and calculate an average of these values
- After 5 minutes (300 seconds) switch off the power supply and stop the stop-clock
- Now measure the mass of the vacuum flask and water once more, and calculate how much the mass has decreased by. This will give the mass of water that has changed state (which you will use in determining your result)
- The heat supplied to the material can be calculated using the equation:
energy = current × voltage × time
(Note: the time must be in seconds)
- These values, along with the change in mass, can now be substituted into the top equation to find the specific latent heat for steam
- A similar experiment can be carried out for ice (in a beaker this time), but the ice must be measured separately from the beaker otherwise you’ll be measuring the mass of the water as well
- It is also important to ensure that the temperature of the ice is 0 °c at the start of the experiment. This will be the case if the ice has started to melt