Plastics & Man-Made Fibres
Plastics, nylon and terylene
- These are synthetic polymers with many uses
- Nylon is a copolymer used to produce clothing, fabrics, nets and ropes
- Terylene is a polyester made from monomers which are joined together by ester links
- Terylene is used extensively in the textile industry and is often mixed with cotton to produce clothing
- Synthetic polymerisation also produces plastics that have many different uses in today’s society
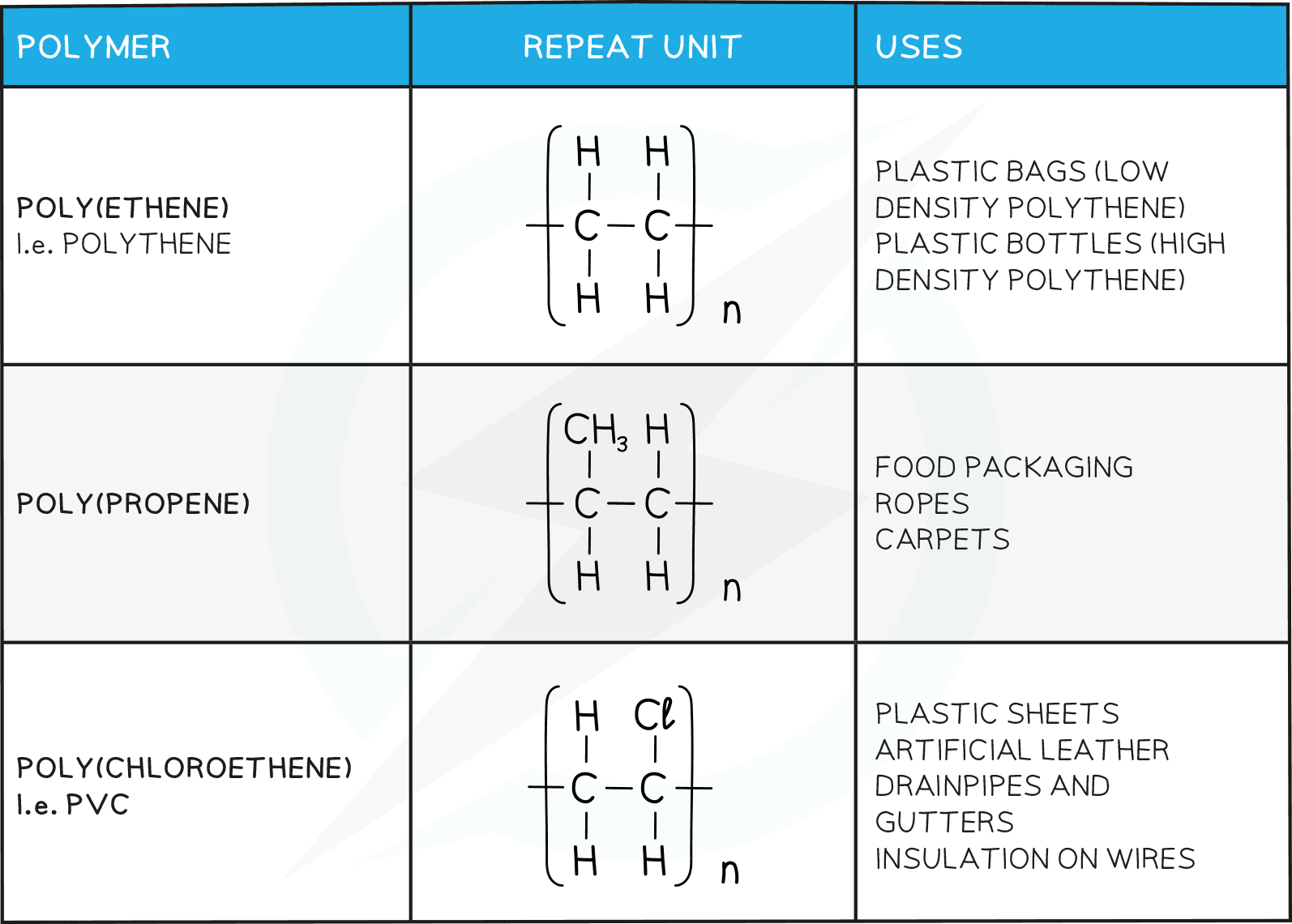
Uses of plastics
Non-biodegradable plastics
- These are plastics which do not degrade over time or take a very long time to degrade, and cause significant pollution problems
- In particular plastic waste has been spilling over into the seas and oceans and is causing huge disruptions to marine life
- In landfills waste polymers take up valuable space as they are non-biodegradable so microorganisms cannot break them down. This causes the landfill sites to quickly fill up
- Polymers release a lot of heat energy when incinerated and produce carbon dioxide which is a greenhouse gas that contributes to climate change
- If incinerated by incomplete combustion, carbon monoxide will be produced which is a toxic gas that reduces the capacity of the blood to carry oxygen
- Polymers can be recycled but different polymers must be separated from each other which is a difficult and expensive process
Extended Only
Addition & Condensation Polymers & Deducing Structures
Addition polymerisation
- Addition polymers are formed by the joining up of many monomers and only occurs in monomers that contain C=C bonds
- One of the bonds in each C=C bond breaks and forms a bond with the adjacent monomer with the polymer being formed containing single bonds only
- Many polymers can be made by the addition of alkene monomers
- Others are made from alkene monomers with different atoms attached to the monomer such as chlorine or a hydroxyl group
- The name of the polymer is deduced by putting the name of the monomer in brackets and adding poly- as the prefix
- For example if propene is the alkene monomer used, then the name is polypropene
Examples of addition polymerisation: polythene and PVC
Condensation polymerisation
- Condensation polymers are formed when monomer molecules are linked together with the removal of a small molecule, usually water
- Condensation polymerisation usually involves two different monomers, each one having a functional group on each end
- Hydrolysing (adding water) to the compound in acidic conditions usually reverses the reaction and produces the monomers by rupturing the peptide link
Condensation produces the polyamide which is ruptured at the link by hydrolysis in the reverse reaction
Deducing the monomer from the polymer
- Polymer molecules are very large compared with most other molecules
- Repeat units are used when displaying the formula:
- Change the double bond in the monomer to a single bond in the repeat unit
- Add a bond to each end of the repeat unit
- The bonds on either side of the polymer must extend outside the brackets (these are called extension or continuation bonds)
- A small subscript n is written on the bottom right-hand side to indicate a large number of repeat units
Diagram showing the concept of drawing a repeat unit of a monomer
Deducing the polymer from the monomer
- Identify the repeating unit in the polymer
- Change the single bond in the repeat unit to a double bond in the monomer
- Remove the bond from each end of the repeat unit and the subscript n
Diagram showing how to deduce the structure of a monomer from a repeat unit
Example: Deducing the structure of chloroethene from a repeat unit of Poly(chloroethene)
Diagram showing the monomer from the repeat unit of an addition polymer (polychloroethene)
Formation of nylon
- Nylon is a polyamide made from dicarboxylic acid monomers (a carboxylic with a -COOH group at either end) and diamines (an amine with an -NH2 group at either end)
- Each -COOH group reacts with another -NH2 group on another monomer
- An amide linkage is formed with the subsequent loss of one water molecule per link
The condensation reaction in which the polyamide Nylon is produced
Formation of terylene
- Terylene is a polyester made from dicarboxylic acid monomers (a carboxylic with a -COOH group at either end) and diols (an alcohol with an -OH group at either end)
- Each -COOH group reacts with another -OH group on another monomer
- An ester linkage is formed with the subsequent loss of one water molecule per link
The condensation reaction in which the polyester Terylene is produced
Exam Tip
You should be able to draw the box diagrams representing polymers where each box represents a part of the repeating hydrocarbon chain.
The functional groups on the monomers and the link formed in the polymers are the important parts and must be clearly drawn.