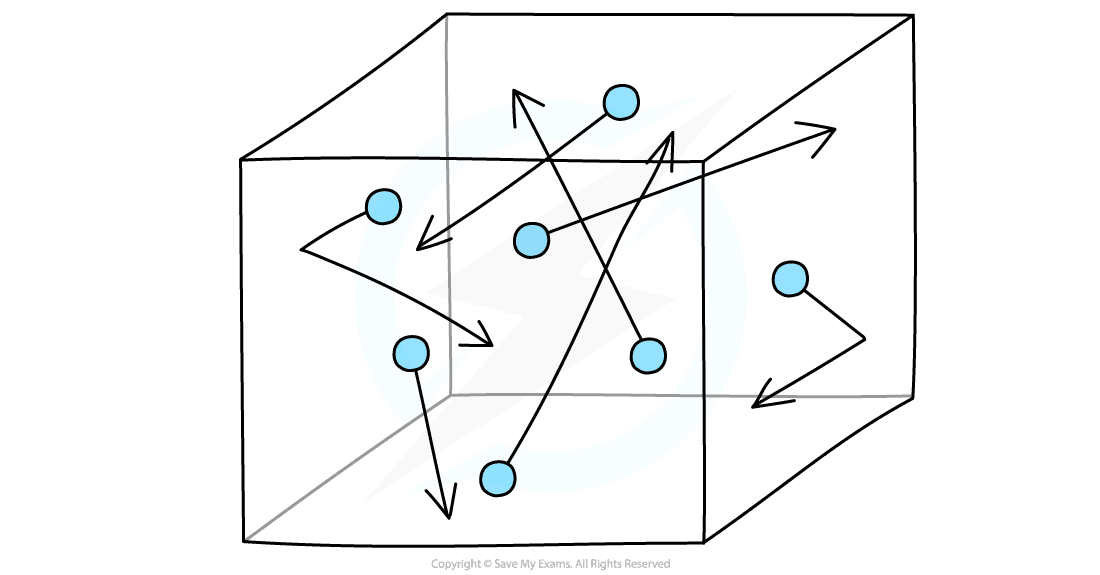
Motion of Molecules in a Gas
- The molecules in a gas move around randomly at high speeds
- The temperature of a gas is related to the average speed of the molecules:
The hotter the gas, the faster the molecules move

Gas molecules move about randomly at high speeds
- As the molecules move around, they collide with the surface of nearby walls
- Each collision applies a force across the surface area of the walls
- Pressure is the force per unit area:

and hence a pressure will be exerted on those wallsExtended Only
Molecule Momentum
- When molecules collide against a wall, they bounce off, changing their momentum
When molecules (in a gas) collide against a wall, they undergo a change in momentum
- There are many such collisions every second, resulting in a large change in momentum each second
- This change in momentum each second results in a force being exerted against the wall:

- The pressure exerted by the gas is equal to this force divided by the area of the wall
Brownian Motion
- When small particles (such as pollen or smoke particles) are suspended in a liquid or gas, the particles can be observed through a microscope moving around in a random, erratic fashion
Brownian Motion: the erratic motion of small particles when observed through a microscope
- This movement is called Brownian Motion
- This motion is caused by molecules in the gas (or liquid) colliding at high speeds with the small particles
Gas molecules move at high speeds, collide with larger particles, giving them a little nudge
- These collisions give the particles a little nudge, causing them to change their speed and directions randomly, each time they are struck by a molecule
- This effect provides important evidence concerning the behaviour of molecules in gases
Extended Only
Massive Particles Moved by Small Molecules
- The small particles observed in Brownian motion are significantly bigger than the molecules that cause the motion
- The molecules are able to affect the particles in this way because they are travelling at very high speeds (much higher than the particles) and so have a lot of momentum, which they transfer to the particles when they collide