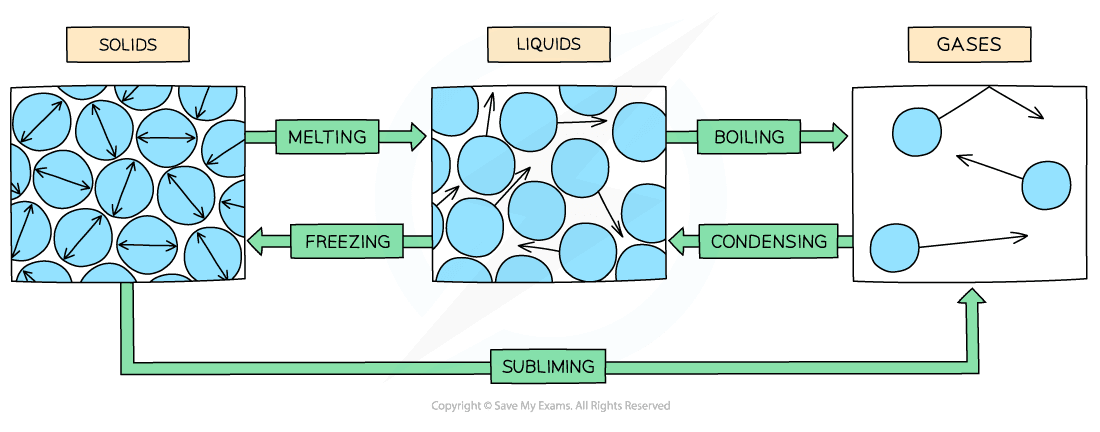
Arrangement & Motion of Molecules
Diagram showing the arrangement and motion of different states of matter
- In a solid:
- The molecules are very close together and arranged in a regular pattern
- The molecules vibrate about fixed positions
- In a liquid:
- The molecules are still close together (no gaps) but are no longer arranged in a regular pattern
- The molecules are able to slide past each other
- In a gas:
- The molecules are widely separated – about 10 times further apart in each direction
- The molecules move about randomly at high speeds
Extended Only
The Forces & Distances between Molecules
- In a solid:
- The molecules are held in place by strong intermolecular bonds
- These bonds prevent the molecules from moving, giving the solid its rigid shape and fixed volume
- In a liquid:
- The molecules have enough energy that they are able to break the bonds between them
- The bonds are still there, but they no longer hold the molecules in place
- As a result, the molecules can move around (by sliding past each other) allowing the liquid to flow
- In a gas:
- The molecules are now moving around randomly at high speeds
- The molecules have broken the bonds between them: They are widely separated with no long-range forces binding them together
- As a result the molecules are able to move freely and so the gas can flow freely
- Because of the large spaces between the molecules (along with the absence of long-range forces) the gas can easily be compressed and is also able to expand