Alpha Particle Scattering
- Evidence for the structure of the atom comes from the study of α-particle scattering
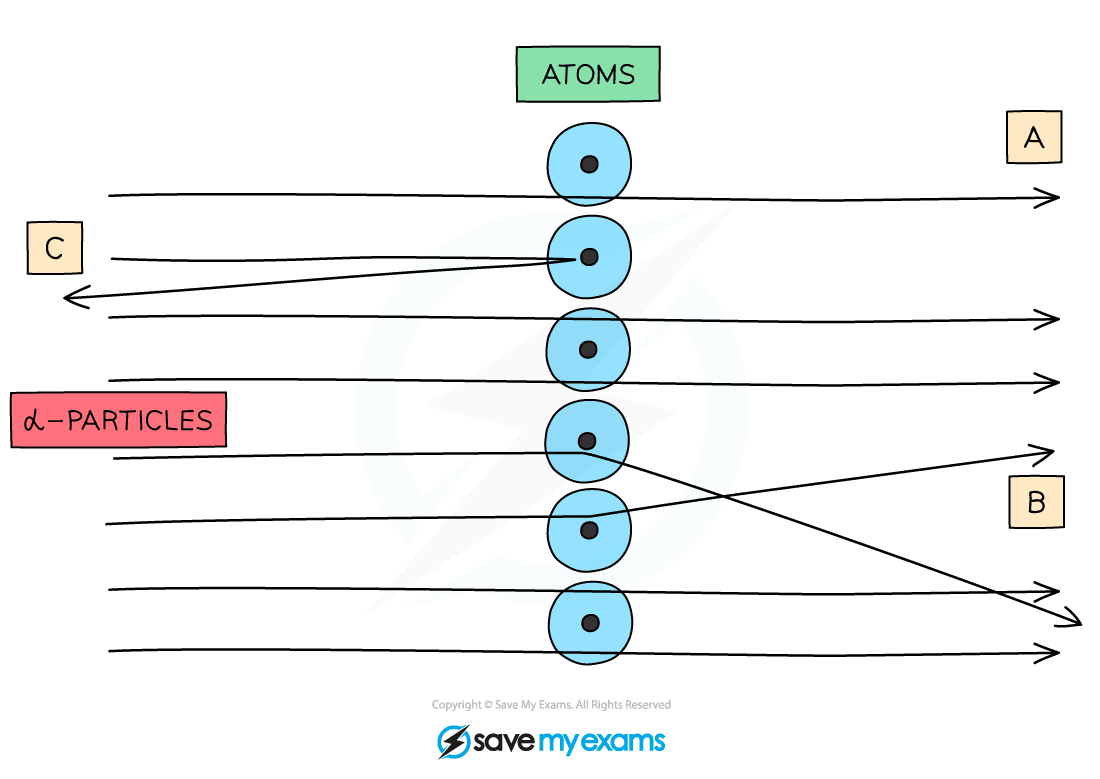
When α-particles are fired at thin gold foil, most of them go straight through but a very small number bounce straight back
- When α-particles are fired at thin pieces of gold foil:
- The majority of them go straight through (A)
This happens because the atom is mainly empty space - Some are deflected through small angles (B)
This happens because the positive α-particles are repelled by the positive nucleus - A very small number are deflected straight back (C)
This is because the nucleus is extremely small
- The majority of them go straight through (A)