The Nature of Decay
- Radiation consists of high energy particles (or waves) emitted from the nucleus of an unstable atom

Unstable nuclei decay by emitting high energy particles – radiation
- There are three (main) types of radiation: alpha (α) particles, beta (β−) particles, or gamma (γ) rays
- Radiation is emitted randomly
This means that, although we understand why some nuclei emit radiation, it is impossible to predict exactly when a nucleus will emit radiation
The Properties of Radiation
- Alpha (α) particles are high energy particles made up of 2 protons and 2 neutrons (the same as a helium nucleus).
They are usually emitted from nuclei that are too large - Beta (β−) particles are high energy electrons emitted from the nucleus (even though the nucleus does not normally contain any electrons)
They are usually emitted by nuclei that have too many neutrons - Gamma (γ) rays are high energy electromagnetic waves
They are emitted by nuclei that need to lose some energy - If these particles hit other atoms, they can knock out electrons, ionising the atom
When radiation passes close to atoms, it can knock out electrons, ionising the atom
- Ionisation can cause chemical changes in materials, and can damage or kill living cells
The nature and properties of the different types of radiation are summarised in the table below
- Note that when you go down the table, the range and penetration increase, but the ionisation decreases
Diagram showing the penetrative abilities of alpha (α) particles, beta (β−) particles, and gamma (γ) rays. Note how some of the gamma rays are able to penetrate the leadhttps://player.vimeo.com/video/374078109?title=0&byline=0&portrait=0Extended Only
Deflection in Electric & Magnetic Fields
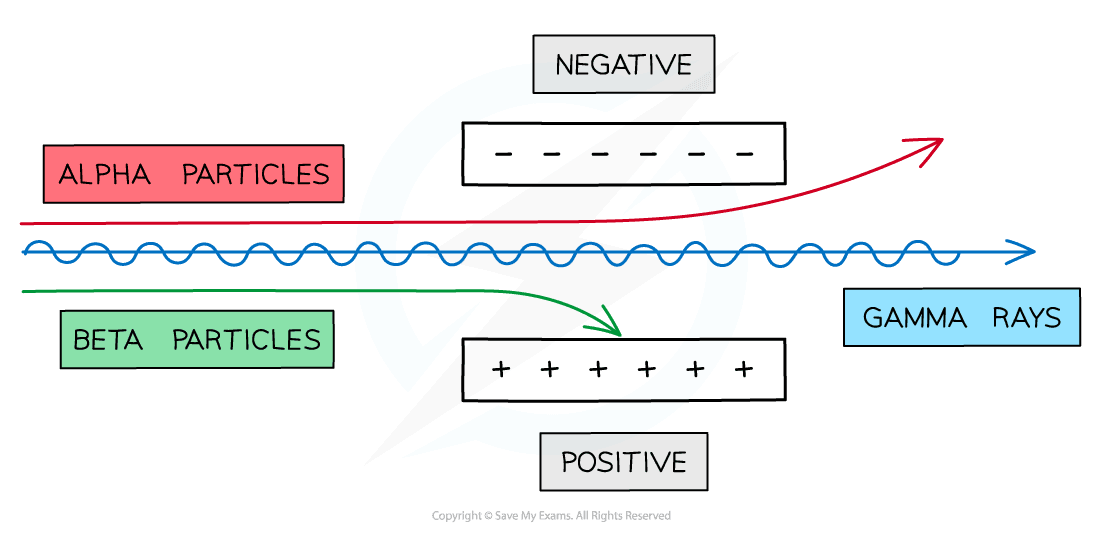
- Because of their charges, alpha and beta particles can be deflected by electric and magnetic fields
Alpha and Beta particles can be deflected by electric fields
- Because they have opposite charges, alpha and beta particles are deflected in opposite directions
- Beta is deflected by more than alpha, because beta particles have a much smaller mass
- Gamma is not deflected because gamma rays have no charge
Alpha and Beta particles can also be deflected by magnetic fields
Diagram showing an experiment to find the type of radiation being emitted by a sourceExtended Only
Ionisation
- Alpha is by far the most ionising form of radiation
- Alpha particles leave a dense trail of ions behind them, affecting virtually every atom they meet
- Because of this they quickly lose their energy and so have a short range
- Their short range makes them relatively harmless if handled carefully, but they have the potential to be extremely dangerous if the alpha emitter enters the body
- Beta particles are moderately ionising
- The particles create a less dense trail of ions than alpha, and consequently have a longer range
- They tend to be more dangerous than alpha because they are able to travel further and penetrate the skin, and yet are still ionising enough to cause significant damage
- Gamma is the least ionising form of radiation (although it is still dangerous)
- Because Gamma rays don’t produce as many ions as alpha or beta, they are more penetrating and have a greater range
- This can make them hazardous in large amounts
Extended Only
Applications of Radioactivity
- Radioactivity has a large number of uses in both medicine and industry, some of which are listed below
Measuring the thickness of materials:
Beta particles can be used to measure the thickness of thin materials such as paper, cardboard or aluminium foil
- As a material moves above a beta source, the particles that are able to penetrate it can be monitored using a detector
- If the material gets thicker more particles will be absorbed, meaning that less will get through
- If the material gets thinner the opposite happens
- This allows the machine to make adjustments to keep the thickness of the material constant
- Note: Devices like this use beta radiation because it will be partially absorbed by the material
If alpha particles were used all of them would be absorbed and none would get through
If gamma were used almost all of it would get through and the detector would not be able to sense any difference if the thickness were to change
Tracers
- Tracers are radioactive isotopes that can be added to some fluid so that the flow of that fluid can be monitored
- Tracers have numerous uses in both medicine and industry:
- In medicine tracers can be added to the blood to check blood flow around the body and search for blockages (blood clots)
- In industry tracers may be added into an oil pipeline in order to check for any leaks
- In all cases:
- The amount used is kept to a minimum to reduce people’s exposure to radiation
- Isotopes are chosen that have short half-lives of around a few hours: long enough to carry out the procedure, but not so long that they cause long-term harm
- Gamma radiation is used as it is highly penetrating (you can detect it) and low ionising (minimising harm)
Radiotherapy
- Radiotherapy is the name given to the treatment of cancer using radiation
(Chemotherapy is treatment using chemicals) - Although radiation can cause cancer, it is also highly effective at treating it
- Radiation can kill living cells. Some cells, such as bacteria and cancer cells, are more susceptible to radiation than others
- Beams of gamma rays are directed at the cancerous tumour
(Gamma rays are used because they are able to penetrate the body, reaching the tumour) - The beams are moved around to minimise harm to healthy tissue whilst still being aimed at the tumour
Sterilisation
- Medical instruments are sterilised by exposing them to gamma rays
- The gamma rays kill bacteria on the instruments and destroy viruses
- Gamma rays are far more effective at killing bacteria than either boiling water or chemical treatment and are able to penetrate the instruments reaching areas that may otherwise not be properly sterilised