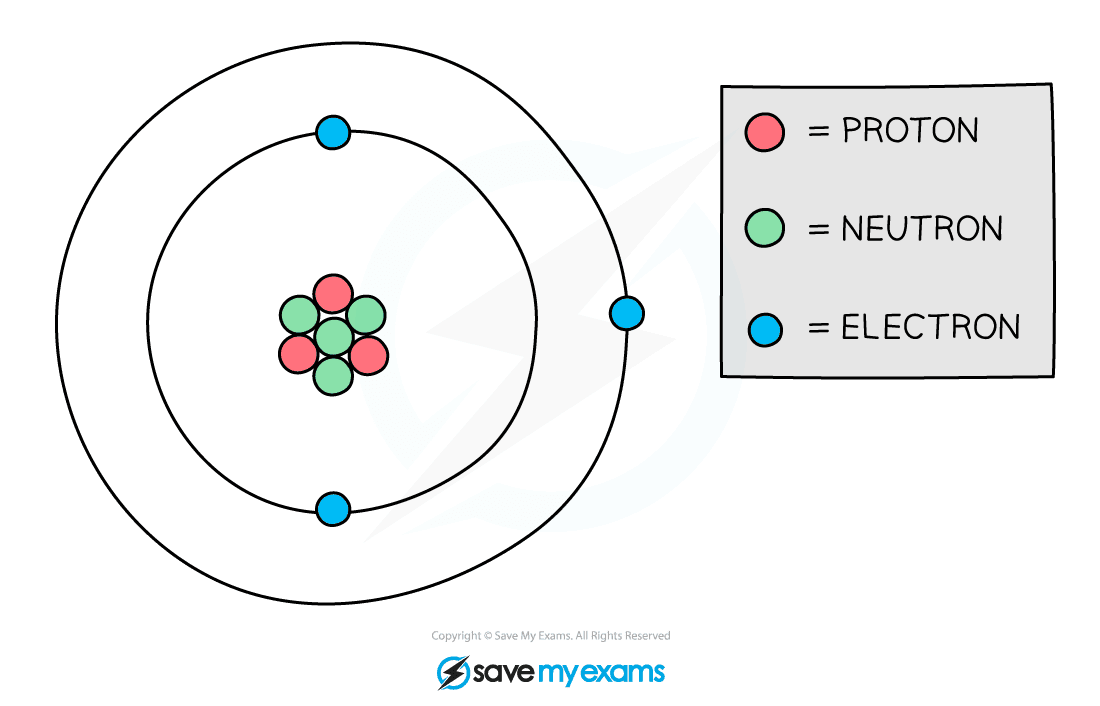
Protons & Neutrons
- Atoms are made up of three different particles:
Protons and neutrons are found in the nucleus of an atom
The properties of each of these particles is shown in the table below:
- The atom shown in the above diagram can also be represented using an atomic symbol:

Atomic symbols, like the one above, describe the constituents of nuclei
- The top number is called the nucleon number, A, and is equal to the total number of particles (protons and neutrons) in the nucleus
- The lower number is called the proton number, Z, and is equal to the total number of protons in the nucleus
(Note: Chemists refer to the nucleon number as the mass number, and the proton number as the atomic number)
- When given an atomic symbol, you can figure out the total number of protons, neutrons and electrons in the atom:
- Protons: The number of protons is equal to the proton number
- Electrons: Atoms are neutral, and so in a neutral atom the number of negative electrons must be equal to the number of positive protons
- Neutrons: The number of neutrons can be found by subtracting the proton number from the nucleon number
- The term nucleon is used to mean a particle in the nucleus – ie. either a proton or a neutron
- The term nuclide is used to refer to a nucleus with a specific combination of protons and neutrons
Isotopes
- Although all atoms of the same element always have the same number of protons (and hence electrons), the number of neutrons can vary:
The three atoms shown above are all forms of hydrogen, but they each have different numbers of neutrons
- The number of neutrons in an atom does not affect the chemical properties of an atom, only its mass. Such atoms are called isotopes:
Isotopes are atoms (of the same element) that have equal numbers of protons but different numbers of neutrons