Methods of Purification
The choice of the method of separation depends on the nature of the substances being separated. All methods rely on there being a difference of some sort, usually in a physical property such as b.p., between the substances being separated.
Mixtures of solids
- Differences in density, magnetic properties, sublimation and solubility can be used
- For a difference in solubility, a suitable solvent must be chosen to ensure the desired substance only dissolves in it and not other substances or impurities
Mixtures of liquids
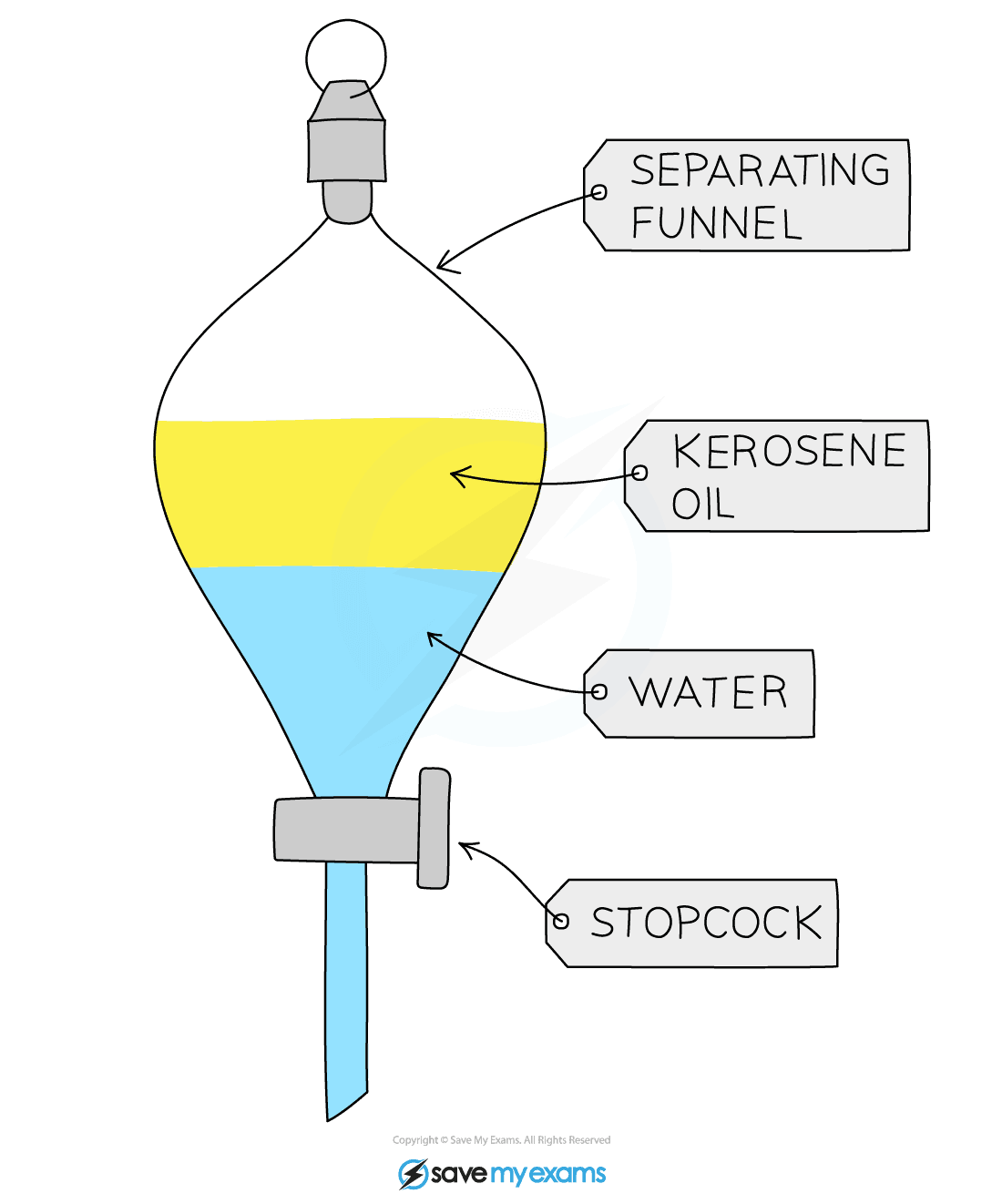
- Immiscible liquids can be separated using a separating funnel or by decanting (pouring carefully)
- Examples include when an organic product is formed in aqueous conditions
Separating funnel being used to separate kerosene and water
Filtration
- Used to separate an undissolved solid from a mixture of the solid and a liquid / solution ( e.g. sand from a mixture of sand and water). Centrifugation can also be used for this mixture
- Filter paper is placed in a filter funnel above another beaker
- Mixture of insoluble solid and liquid is poured into the filter funnel
- Filter paper will only allow small liquid particles to pass through as the filtrate
- Solid particles are too large to pass through the filter paper so will stay behind as a residue
Filtration of a mixture of sand and water
Crystallisation
- Used to separate a dissolved solid from a solution, when the solid is much more soluble in hot solvent than in cold (e.g. copper sulphate from a solution of copper (II) sulphate in water)
- The solution is heated, allowing the solvent to evaporate to leave a saturated solution behind
- Test if the solution is saturated by dipping a clean, dry, cold glass rod into the solution. If the solution is saturated, crystals will form on the glass rod
- The saturated solution is allowed to cool slowly and solids will come out of the solution as the solubility decreases, and crystals will grow
- Crystals are collected by filtering the solution
- They are then washed with cold, distilled water to remove impurities and allowed to dry
Diagram showing the process of crystallisation
Simple Distillation
- Used to separate a liquid and soluble solid from a solution (e.g. water from a solution of saltwater) or a pure liquid from a mixture of liquids
- The solution is heated and pure water evaporates producing a vapour which rises through the neck of the round-bottomed flask
- The vapour passes through the condenser, where it cools and condenses, turning into pure liquid H2O which is collected in a beaker
- After all the water is evaporated from the solution, only the solid solute will be left behind
Diagram showing the distillation of a mixture of salt and water
Fractional distillation
- Used to separate two or more liquids that are miscible with one another (e.g. ethanol and water from a mixture of the two)
- The solution is heated to the temperature of the substance with the lowest boiling point
- This substance will rise and evaporate first, and vapours will pass through a condenser, where they cool and condense, turning into a liquid that will be collected in a beaker
- All of the substance is evaporated and collected, leaving behind the other components(s) of the mixture
- For water and ethanol: ethanol has a boiling point of 78 ºC and water of 100 ºC. The mixture is heated until it reaches 78 ºC, at which point the ethanol boils and distils out of the mixture and condenses into the beaker
- When the temperature starts to increase to 100 ºC heating should be stopped. Water and ethanol are now separated
Fractional distillation of a mixture of ethanol and water